When calcium aluminate cement is added to Portland Cement the hardening acceleration mechanism changes. Mixtures made only with Portland cement may be inadequate in terms of offering solutions to various problems such as high temperatures, early strength, rapid setting and preventing cracks. This is why binding systems referred to as ternary binding systems are created using different ratios of Portland cement, calcium aluminate cement and calcium sulfate (gypsum). The mixing ratios in a ternary system can be altered to offer solutions to various needs such as adjustable setting time, development of early strength, rapid setting and prevention of contraction cracking.
Portland Cement Hydration
Portland cement typically consists of mineralogical phases in the interval amounts specified below:
- Alite (C3S, 40-70%)
- Belite (C2S, 10-40%)
- Aluminate (C3A, 1-15%)
- Ferrite (C4 AF, 1-15%)
- Free lime (CaO, 0.5-3%)
The aluminate phase is a very reactive phase. This is why the sudden hardening we call “Flash Set” occurs when it comes into contact with water. The hydration reactions of the aluminate phase are slowed down and controlled with finely ground intermediate products of clinker and gypsum.
Calcium Aluminate Cement Hydration
Calcium aluminate cement consists of mineralogical phases in the interval amounts specified below:
- Calcium monoaluminate (CA, 50-55%)
- Mayenite (C12A 7, 1-3%)
- Gehlenite (C2 AS, 1-15%)
- Ferrite (C4 AF, 15-25%
The aluminate phase is very reactive, so setting occurs rapidly when it comes into contact with water. When grinding calcium aluminate cement clinker, no additives are used.
How Calcium Aluminate Cement Affects Setting Time
Gypsum coats the surface of the C3A phase, thus preventing the instant reaction of this phase and ensuring a more measured setting time. The amount of calcium aluminate cement that must be added in quick-setting applications varies depending on the type of gypsum in the Portland cement. This amount is high in the presence of gypsum and hemihydrate, and low in the presence of anhydrous gypsum. When calcium aluminate cement is added, the sulfate ions (SO42-) found in the gypsum, combine with the aluminate ions released by the calcium aluminate cement to form ettringite instead of the C3A phase in Portland cement. When a sufficient amount of calcium aluminate cement is added, it reacts with C3A, and no sulfate remains available to limit this reaction. In this situation, a quick setting characteristic is obtained due to C3A phase hydration. The ettringite that forms contributes to early strength.

Figure 1. SEM Image of Ettringite
Calcium Aluminate Cement Additive System

Figure 2. Graphic of Heat Generated Over Time
According to the Iso-calorimetry results provided in the figure,
- In systems where calcium aluminate cement is predominant, there is a single extreme peak due to the CA phase reaction.
- In systems where Portland cement predominates, there are three different minor peaks. The reason for this is the delay in the formation of ettringite.
Amount of Calcium Aluminate Cement Additive and Gypsum: Experiments
How the amount of calcium aluminate cement affects Portland cements with different types of gypsum.
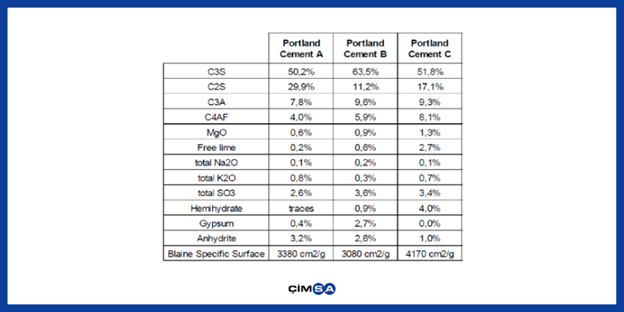
Figure 3. Composition and Properties of Cement Used in Different Products
Product A:
A small amount of calcium aluminate cement is used because anhydrous gypsum is the main type.
Product C:
A large amount of calcium aluminate cement is used because hemihydrate gypsum is the main type.
Product B:
A moderate amount of calcium aluminate cement is used in the other two products because they contained both anhydrite and gypsum.

Figure 4. Changes in Setting Time Depending on the Amount of Calcium Aluminate Cement Added to Cements with Different Compositions
As depicted in the graphic above, for a rapid 10-minute setting time:
- Percentage of calcium aluminate cement used in Product A is 14%
- Percentage of calcium aluminate cement used in Product B is 27%
- Percentage of calcium aluminate cement used in Product C is 37%
The Hardening Acceleration Mechanism in Portland Cement Achieved by Adding Calcium Aluminate Cement
When calcium aluminate cement is added to Portland Cement, the hardening acceleration mechanism changes. As a result, the necessary solutions to problems such as high temperature, early strength, rapid setting and cracking can be obtained by altering the recipe in ternary systems. This experiment demonstrated:
- The dissolution rate of calcium aluminate cement is higher than anhydrite and lower than gypsum and hemihydrate.
- When using Portland cement that contains anhydrite or when the type of calcium sulfate used is anhydrous, very little should be added because the solubility of calcium aluminate cement is more rapid than anhydrite.
- In situations where the dissolution of calcium aluminate cement is higher, needle-shaped ettringite structures form.
- When Portland cement containing gypsum (calcium sulfate type gypsum) or hemihydrate is used, a lot of calcium aluminate cement is required because its solubility rate is lower than both of these types of gypsum.
- In both of these situations, the calcium aluminate cement consumes the sulfate ions and prevents the C3A phase reaction.
- In situations where the gypsum solubility is greater, shorter ettringite structures form. The ettringite that forms, however, contributes to early strength.
References Used:



